VI. Disposal of high level wastes
Zharikov V.A. and Zaraisky G.P. Complex experimental studies concerning the disposal of radioactive wastes in abyssal geological formations
First experimental data were obtained on the interaction of radioactive wastes (RAW) with rocks and underground waters at high temperatures. Results obtained allow the more precise forecast of RAW behavior during their long-term concervation in the earth's interior. Both negative and positive effects of increasing temperature were definitely proved: transport processes become more intense, but binding radionuclides by surrounding rocks is also enhanced as a result of increasing rates of chemical reactions. First promising results were obtained as to the significant reduction of rock permeability by means of filtration of water through the fractures at low-temperature conditions (below 250oC). This process results in sealing fractures by newly formed minerals (Zharikov et al., 1994).
The stability of industrial Na-Al-phosphate glass with radioactive impurities (90Sr and 137Cs) in "dry"system and in contact with water was studied by topologic and thermodynamic analysis. The dry supercooled glass was shown to be unstable below 500oC and decompose to sodium orthophosphate + sodium metaphosphate + berlinite + Sr-whitlockite. MAGATE tests of glass solubility are not sufficient to evaluate the safety of the disposal of solid RAW (Mayak Production Association); the solubility of minerals resulting from the glass recrystallization should also be studied (Ivanov et al., 1994).
It was experimentally established that Na-Al-phosphate glass is unstable in contact with water at 200 and 300oC and completely recrystallizes in a few days. In this process, Sr is bound by newly formed phosphates, while Cs almost fully passes into the aqueous solution. Different rocks (granite, gabbro, dunite) were found to have different ability to bind Cs and Sr leached by water from the RAW-glass. Thus, silica-rich rocks (granite) are favorable for Sr storage, whereas ultrabasic rocks (porphyrite, gabbro, dunite) are preferable for Cs storage (Shapovalov, 1994).
Experiments on sorption of Sr on porphyrite were performed at 250oC and 200 bar by means of the diffusion of porous chloride solution. Sr was absorbed by pophyrite at the beginning of the column, where Sr content was maximum (about 4%). Thus, at high temperatures a great quantity of Sr can be retained just within the nearest zones of porphyrite.
Zyryanov V.N., Kotelnikov A.R., and Suvorova V.A. Immobilization of radionuclides in crystalline composite materials.
1. The conception of phase and chemical correspondence was applied to the rock and mineral matrices suitable for the long-term immobilization of high level waste (HLW). The studies involved the evaluation of mineralogical and geochemical features of liquid HLW of nuclear industry and igneous rocks of some natural complexes. This approach offers the following opportunities:
-optimal choice of mineral capable of concentrating HLW;
-determination of the matrix type (synthetic rock);
-determination of geochemical, mineralogical, and petrological criteria for the choice of natural rock
41
complexes for the long-term disposal of matrices with HLW;
-minimization of the radionuclide migration to the natural environment.
The geochemical evaluation of a number of magmatic complexes of the Earth's crust was performed to reveal the objects suitable for HLW disposal. Agpaitic ultraalkaline rocks (Lovozero alkaline massif, Kola Peninsula, and Ilimaussak massif, Greenland) were shown to correspond most closely to the immobilizing matrices, judging by their geochemical features (Zharikov et al., 1994; Kotelnikov et al., 1994).
2. A wide variety of natural and synthetic minerals promising for HLW disposal were studied. Synroc, titanate-zirconate, aluminosilicate, and Zr-phosphate samples incorporating Cs, Sr, Ce, Eu, and I were synthesized by hot pressing and pyrosynthesis. The imitator element contents in these samples were close to the limiting values (theoretically tolerable). The MCC-1 and Soakslet tests (for leach rate) showed the much higher stability of the mineral matrices compared to borosilicate glasses, which are used in nuclear industry for the immobilization of HLW (Orlova et al., 1993; Akhmedzhanova et al., 1993).
Promising matrix materials suitable for the immobilization of Cs and Sr were synthesized on the basis of sodium Zr-phosphates (NZP) by single-axis and isostatic hot pressing at 1200oC and 25 MPa. This method provides the high-density ceramics (95% of theoretical value) that excels Synroc in chemical stability (Orlova et al, 1993).
Crystalline matrices suitable for immobilization and safe storage of transuranium elements were synthesized on the basis of the NZP ceramics incorporating rare-earth actinide imitators (Gd, Nd, Y). The samples obtained outperform Synroc by a factor of 102-103 in resistance to leaching.
Synthetic rocks (model granites and nepheline syenites) with Cs- and Sr-bearing minerals were obtained by ceramization of glass at 600-750oC and 1 kbar. As much as 90% of theoretical rock density was reached.
3. The distribution of isomorphic components between rock-forming silicates and fluid was studied by cation-exchange equilibrium methods. The distribution of alkaline and alkaline-earth elements between feldspar and brine fluid was studied in the NaAlSi3O8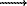 SrAl2Si2O8, KAlSi3O8
SrAl2Si2O8, KAlSi3O8 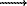 SrAl2Si2O8, and KAlSi3O8
SrAl2Si2O8, and KAlSi3O8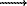 BaAl2Si2O8 binary systems at 700-800oC. The unit cell parameters were determined for the synthesized feldspar solid solutions. Na
BaAl2Si2O8 binary systems at 700-800oC. The unit cell parameters were determined for the synthesized feldspar solid solutions. Na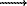 Sr and K
Sr and K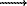 Ba feldspars displayed positive deviations from ideal solutions. K
Ba feldspars displayed positive deviations from ideal solutions. K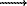 Sr feldspars were found to have negative mixing volumes.
Sr feldspars were found to have negative mixing volumes.
The experiments on the distribution of Sr and Ba among plagioclase, alkaline feldspar, and brine fluid showed that at 400-800oC and 1-2 kbar feldspars are enriched in Sr and Ba relative to the fluid. The contents of these elements in feldspar solid solution ranged up to 30-40 mol%. With increasing temperature, Ba passed from the solution to plagioclase, while Sr showed the opposite behaviour. These data allow to conclude that plagioclase can be used as a matrix for the immobilization of strontium radionuclides.
Chlorine-, bromine-, iodine-. and sulfate-bearing sodalites were synthesized. The anion-exchange rate was shown to decrease in the series Cl-SO4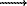 Cl-Br
Cl-Br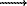 Cl-I. The I-sodalite failed to participate in the ion-exchange reactions and, therefore, can be used for the immobilization of radioactive iodine.
Cl-I. The I-sodalite failed to participate in the ion-exchange reactions and, therefore, can be used for the immobilization of radioactive iodine.
The partition of K and Cs between kalsilite and brine fluid was studied at 800oC and 2 kbar. A miscibility gap was revealed in the (K,Cs)AlSiO4 solid solution, and a nonideal partition of K and Cs between kalsilite and fluid was established.
The leach rates of Na, Al, Si, K, and Ca were determined (MAGATE and MCC-1 tests, 90oC, distilled water) for natural alkaline feldspar, labrador, scapolite, sodalite, nepheline, and apatite which are considered as potential HLW concentrators within a synthetic matrix. Data obtained show that framework aluminosilicates can be used as matrices for radionuclide immobilization along with high-stability ceramic materials (Kotelnikov et al., 1995).
4. The thermodynamic processing of the experimental data on cation-exchange equilibria between Sr- and Ba-feldspars and brine solutions was performed to evaluate the thermodynamic properties of the candidate minerals. The relationship between the integral excess energies of mixing and the difference of ionic radii ( R) of isomorphic elements was derived at 700-800oC and 2 kbar. For the binary feldspar solid solutions with heterovalent isomorphism (Na-Ca, Na-Sr, Na-Ba, K-Ba, K-Sr) and alkaline feldspars with isovalent isomorphism of monovalent cations (Na-Ca, Na-Rb, K-Rb), this relationship is described by the following equation:
R) of isomorphic elements was derived at 700-800oC and 2 kbar. For the binary feldspar solid solutions with heterovalent isomorphism (Na-Ca, Na-Sr, Na-Ba, K-Ba, K-Sr) and alkaline feldspars with isovalent isomorphism of monovalent cations (Na-Ca, Na-Rb, K-Rb), this relationship is described by the following equation:
Gexin=0.578 + 5,690 R - 27.844
R - 27.844 R2 + 68.407
R2 + 68.407  R3
R3
For the binary solid solutions with isovalent isomorthism of divalent cations (Ca-Sr, Ca-Ba, Sr-Ba), this relatonship appears as follows:
Gexin=0,695 + 0.407  R + 0.44
R + 0.44 R2
R2
Such polynomial equations were also obtained for the integral excess volumes of mixing. The correlations between the crystallochemistry and energetic properties of the feldspar solid solutions were established (Kroll et al., 1995; Kotelnikov et al., 1995).
5. Experiments on iodine sorption by NaX- and Cu-bearing NaX-zeolites with the subsequent formation of stable zeolite matrices were performed to solve the problem of immobilization of the molecular iodine resulting from the processing waste fuel at radiochemical plants. The optimal conditions of iodine sorption and zeolite ceramization were determined at T=500-800oC and P=1 kbar. The ceramic matrix is composed of feldspars and I-bearing sodalite in the former case and the quartz and corundum mixture disseminated with CuI in the latter. The leach rates of the matrix elements and iodine were estimated (MCC-1 test; 90oC, distilled water). The high potentials of the ceramics as to the radiogenic iodine disposal were proved (Suvorova et al., 19961 , 19962).
A method for the immobilization of Sr and Cs radionuclides was developed on the basis of the ion-exchange sorption of these elements by NaX- and NaA-zeolites with
42
their subsequent transformation into ceramics (feldspars and feldspathoids). The study of the rates of leaching alkaline and alkaline-earth elements from these ceramics (MCC-1 test) showed their stability, which is comparable with Synroc-C samples (Akhmedzhanova et al.,1992; Kotelnikov et al., 1995) .
References:
- Zharikov V.A., Epelbaum M.B., Kotelnikov A.R., and Zaraiskii G.P. (1994) The potential of experimental mineralogy in solving problems of reprocessing and disposal of high-level waste (HLW).// Geochemical problems concerning the disposal of radioactive waste, Essays on Physicochemical Petrology, Iss.18, Miass, pp.6- 16.
- Kotelnikov A.R., Zyryanov V.N., and Epelbaum M.B. (1994) The conception of phase and chemical correspondence applied to the problem of disposal of high-level waste in the earth's crust. // Geochemical problems concerning the disposal of radioactive waste, Essays on Physicochemical Petrology, Iss.18, Miass, pp.83-103.
- Kotelnikov A.R., Zyryanov V.N., and Epelbaum M.B. (1994) Phase and chemical compatibility of matrix minerals and wall rocks at disposal of high-level wastes. // Experiment in GeoSciences, V.3, N.2, pp.9-21.
- Orlova A.I., Zyryanov V.N., Kotelnikov A.R., Demarin T.V., and Rakitina E.V. (1993) Ceramic phosphate matrices for high-level wastes: behaviour under hydrothermal conditions. // Radiokhimiya, V.35, N.6, pp.120-126.
- Akhmedzhanova G.M., Kotelnikov A.R., and Parmuzina M.S. (1993) The study of leach rates of alkaline and alkaline-earth elements from natural minerals (framework aluminosilicates and apatite). // Nuclear Energy and Human Safety NE-83. The IVth Annual Scientific and Technical Conference of the Nuclear Society, June 28-July 2. Abstracts. Nizhnii Novgorod, V.2, pp.965-967.
- Kotelnikov A.R., Akhmedzhanova G.M., and Zyryanov V.N. (1995 ) The stability studies of natural and synthetic minerals applicable as matrices for the immobilization of HLW elements. // The XIII All-Russian Meeting on Experimental Mineralogy , Sept.12-15. Abstracts. Chernogolovka, p.258.
- Kroll H., Kotelnikov A.R., Goettlicher J., Valyashko E.V. (1995) (K,Sr)- feldspar solid solutions: the volume behaviour of heterovalent feldspars. // Eur.J.Mineral., V.7, pp.489-499.
- Kotelnikov A.R. and Chernysheva I.V. (1995) Excess free energies of mixing of Sr,Ba- bearing feldspar solid solutions (experimental data). // Mineralogical Mag., V.59, pp. 79-91
- Suvorova V.A., Akhmedzhanova G.M., Zyryanov V.N. Kotelnikov A.R. and Nekrasov A.N. (1996) Synthesis and properties of iodine-fixing ceramic matrices based on Cu-substituted zeolites. // Experiment in Geoscience, V.5, N.2, pp.49-50.
- Suvorova V.A., Akhmedzhanova G.M., Zyryanov V.N., Kotelnikov A.R., and Tikhomirova V.I. (1996) Study of possible combining radioactive iodine by ceramic matrices based on NaX zeolite. // Experiment in Geoscience, V.5, N.2, pp.50-52.
- Kotelnikov A.R., Bychkov A.M., Zyryanov V.N., Akhmedzhanova G.M.,and Gavlina O.T. ( 1995) The zeolite-feldspar phase transformation as a method of synthesis of aluminosilicate matrices for radionuclide immobilization. // Geokhimiya, N.10, pp.1527-1532.
- Akhmedzhanova G.M., Bychkov A.M., Gavlina O.T., Zyryanov V.N., and Kotelnikov A.R. ( 1995) A method of the solid phase immobilization of radionuclides of alkaline-earth elements, Patent N.2038637, Russian Federation, June 27, 1995, Priority since July 19, 1992, Application N 5057135.
Redkin A.F. Some aspects of the recovery of Cs- and Sr- bearing radioactive wastes.
The investigations were performed with synthetic Na-montmorillonite, analcime and natural clinoptilolites.
1. It was established by experiments that Na-bearing montmorilonites as well as smectites possess high sorption (ion-exchange) capacity relative to Cs (up to 7-10 wt.%), low of Sr one (1-2 wt.%) and practicaly do not sorbe U at 250 - 300oC. In the result of thermodynamic simulation it was shown that Cs and Sr are best to fix (to hold on) in mixed-layer phases in the presence of quartz-muscovite-albite bearing rocks. Pure montmorillonite can not serve as a good sorptive barrier with respect to Cs and Sr (Redkin, 1993).
2. Experimental studies on ion exchange of Na for Cs and Sr in analcime-bearing buffered assemblages were carried out at 250-300oC and P=Psat in pure water and in chloride solution. This investigation showed that such ion exchange of Na for Cs and Sr in analcime changes the Si/Al ratio. The compositions of end members for Cs and Sr analcimes at 250oC are indicated to be Cs0.95Al0.79Si2.17O6 and Sr0.645Al1.41Si1.62O6. Continuous solid solution between Cs and Na does not appear to exist at 250oC and Psat.
Based on the composition of the aqueous chloride solutions, the thermodynamic properties of Cs, Sr, and Na analcimes were determined and used in thermodynamic simulation of Cs and Sr leaching from albite- (Cs, Sr, Na) analcime assemblages at 250oC. In line with the above, the minimum leachability of Cs and Sr from Cs and Sr bearing analcimes is at pH values above 8 with analcime-albite-paragonite buffer. The analcime-albite-paragonite assemblage can therefore act as a potential sorption barrier to the path-way of Cs and Sr migration from HLW owing to the insolubility of Cs and Sr bearing analcimes. These findings have implications to underground HLW repository facilities designed for maximum safety at moderately elevated temperatures (Redkin, Hemley, 1996).
3. Clinoptilolites of three U.S. clinoptilolite-rich tuff deposits, with different alkali and alkaline earth elements mole ratios, were used for experimental investigation. The runs on stability and sorption ability were carried out in air, water and 0.3 molal chloride solution, using individual minerals and using assemblages containing microcline and
43
analcime. The dry experiments were performed up to 1000oC, the hydrothermals from 200 to 450oC, Psat water -1000 bar and run duration from two weeks to 8 months. The clinoptilolites showed different acidic-basic properties, unrelated to exchange capacity, when run under similar conditions. It was also found that Cs-bearing clinoptilolites contained 12-14 wt.% Cs after run in 0.1mNaCl+0.1mKCl+0.1mCsCl, whereas Sr-bearing clinoptilolite contained about 3-5% Sr in 0.1mNaCl+0.1mKCl+0.05mSrCl2. The compositional trend showed some linear variation between Sr and Cs content and Si/Al mole ratio up to Si/Al =5.0. The clinoptilolites rich in bivalence earth alkaline cations possess less thermal stability in air than alkaline enriched clinoptilolites. Hydrothermal investigation showed that (Na-K)-rich clinoptilolite completely decomposes at 325oC, (Na-Mg) rich at 350oC and (Ca-Mg) rich at 375oC in runs of 2 weeks duration in pure water. But, all three natural clinoptilolites were decomposed for 8 months in pure water at 300Ý10oC, Psat.. Nevertheless, we can espouse the Mupton's view that
the upper limit of "stability" of clinoptilolite under hydrothermal conditions is probably about 300oC or a few less.
Na-analcimes associated with clinoptilolites have low Cs and Sr adsorption capacity relative to the clinoptilolites. Na-Sr -bearing analcimes have an inverse dependence of Sr/(Sr+Na+K) mole ratio with Si/Al in the reaction of heterovalent exchange in analcime, where Na and Si are replaced by Sr and Al.
Thermodynamic calculation was done on the solubility of Cs and Sr-bearing clinoptilolites at 300oC, Psat. It was established that under some conditions Sr leachability from Sr-bearing clinoptilolite is insignificant whereas Cs-bearing clinoptilolite leachability is considerably higher (Redkin et al., 1995, 1996).
References:
- Redkin A.F. Investigation of Cs, Sr, U(VI) Sorption on Analcime and Mica-Layer Phases at Elevated T and P. Proceedings of the 1993 Intern. Conf. Nuclear Wast Menag. and Env.Rem. 1993, v.1, pp.163-168.
- Redkin A. F. Hemley J.J., Doughten M. W. and Cygan G.L. The ion-exchange capacity and Cs, Sr leachability of natural clinoptilolites at 200-300oC and Psat. Procceding of Fifth International Conference on Radioactive Waste Management and Environmental Remediation Berlin, Germany, September 3-7, 1995, Vol.1 Cross-Cutting Issues and Management of High-Level Waste and Spent Fuel. ASME, 1995, pp.831-836.
- Redkin A.F., Hemley J.J. Investigation of radionuclide sorption by zeolite-bearing mineral assemblages at elevated temperatures and water pressures. Open-File Report of U.S. Department of the interior of geological survey and Russian Academy of Sciences, 1996, 37p.
Tikhomirova V.I. and Desyatova T.A. Sorption of iodide-ions on porphyrite and schungite.
Within the geoecological program (disposal of radioactive waste (RAW)), we studied the sorption abilities of some rocks that are promising as barriers when RAW storages are eroded by hydrothermal solutions. The sorption of iodide-ions on porphyrite and schungite (rocks quite different in mineral composition) was studied. The potentiometric analysis with a iodine-selective electrode showed the physical adsorption on porphyrite of iodide-ions from electrolyte aqueous solutions; adsorbed ions were readily washed off when the solution was passed through the rock. Schungite, which is 32% amorphous carbon, retained iodide-ions firmly when low-salt solutions were used, and the starting KCl concentration of the model solution decreased by two orders. In the static regime at 25oC, the adsorption isotherm for middle concentrations corresponds to the Freundlich equation. Schungite and activated carbon were compared with respect to the sorption of iodide-ions, and iodine desorption from schungite was studied at 90oC according to MAGATE tests. These studies revealed the correlation between the sorption ability of schungite and the content of the carbonaceous schungite particles on the rock surface, which retains 30% of initially sorbed iodide-ions for a long time by virtue of chemosorption.
Reference:
- Tikhomirova V.I., Desyatova T.A. (1998) The study of the sorption of iodide ions on the barrier rocks of porphyrite and shungite at the burial of RAW. // Geokhimiya, N.2, pp.1-7.
44
Previous
Contents
Next
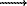 SrAl2Si2O8, KAlSi3O8
SrAl2Si2O8, KAlSi3O8 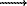 SrAl2Si2O8, and KAlSi3O8
SrAl2Si2O8, and KAlSi3O8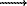 BaAl2Si2O8 binary systems at 700-800oC. The unit cell parameters were determined for the synthesized feldspar solid solutions. Na
BaAl2Si2O8 binary systems at 700-800oC. The unit cell parameters were determined for the synthesized feldspar solid solutions. Na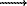 Sr and K
Sr and K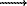 Ba feldspars displayed positive deviations from ideal solutions. K
Ba feldspars displayed positive deviations from ideal solutions. K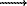 Sr feldspars were found to have negative mixing volumes.
Sr feldspars were found to have negative mixing volumes. R) of isomorphic elements was derived at 700-800oC and 2 kbar. For the binary feldspar solid solutions with heterovalent isomorphism (Na-Ca, Na-Sr, Na-Ba, K-Ba, K-Sr) and alkaline feldspars with isovalent isomorphism of monovalent cations (Na-Ca, Na-Rb, K-Rb), this relationship is described by the following equation:
R) of isomorphic elements was derived at 700-800oC and 2 kbar. For the binary feldspar solid solutions with heterovalent isomorphism (Na-Ca, Na-Sr, Na-Ba, K-Ba, K-Sr) and alkaline feldspars with isovalent isomorphism of monovalent cations (Na-Ca, Na-Rb, K-Rb), this relationship is described by the following equation: